Introduction and Overview: Wettability Testing
The characterization of wettability and surface energy of solid materials has presented challenges to scientists and industrial technicians alike for as long as these attributes have been measured. The fundamental problem is that no direct measurement technique exists: Instead, the vast array of experimental data available is based almost exclusively on the reaction of a liquid or liquids in contact with the solid, usually in the presence of either air or a saturated vapor of the test liquid. Thus, the nature of the test liquid affects the results as surely as the nature of the solid under investigation. Even the gas phase can have a significant effect, though it is usually less pronounced.
Two methods are widely used in science and industry. These are the dyne test, documented in ASTM Std. D2578 and the basis of all ACCU DYNE TEST™ products, which is based on the wettability of calibrated formulations of 2-ethoxyethanol and formamide; and the contact angle test, which relies on measurement of the angle of contact that the test liquid (often reagent grade water) makes on the solid surface. Each has its own advantages, and there is a strong correlation between their results, but it is important to realize that differences in polarity, pH, and solubility parameters of either solid or liquid phase will affect the relationship between the two methods. Additional factors such as the evaporation rate of the liquid, surface topography of the solid, and incorporation of additives or contaminants at the liquid/solid interface will also influence results.
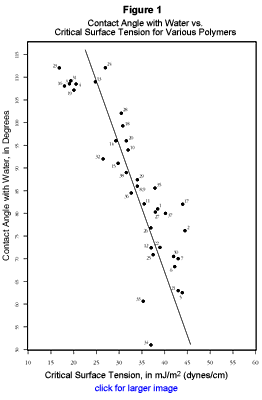
Figure 1 shows the relationship of critical surface tension (data from the dyne test and from Zisman plots, which are based on the contact angles of a homologous series of test liquids of varying surface tension, are both included) to the contact angle of water for a variety of polymers. A data table, along with identification of the 38 polymers included, is available here.In industry, the commonest use of either test is to determine the efficacy of corona, flame, or plasma surface treatment. As long as the treatment is performed in an environment that includes oxygen, any of these methods effectively oxidizes the surface, increasing the polar component of its surface free energy. Generally speaking, the correlation between contact angle and dyne test results will increase as the surface energy of the solid becomes more influenced by polar, as opposed to dispersion, forces.
This means that in most cases there will be very good agreement between the contact angle and dyne solution results for treated polymers. Tables have been published showing this numerical relationship, but we prefer not to present them here—the regression line shown in Figure 1 provides a good estimate of the relationship, and the graphical presentation draws attention to the existence of a real, albeit usually small, systematic variance between the two. As a rule of thumb, the dyne test is best suited to practical testing such as incoming receiving or process control, while the contact angle test is more appropriate for pure research or as an ongoing audit. A number of investigators will find that the effort of employing both methods pays ample dividends.
Whether you are new to surface testing or an experienced hand, we suggest that you review several other pages in this site, which will hopefully clarify and expand on these points. Links are provided in the banner on the right margin, or from the dropdown menus at the top of the page. As always, thanks for reading.
Russell E. Smith, President
April 24, 2024